기사
Prodizen Participates in the Spring Conference of the Korean Society of Nutrition Medicine in 2025
- 작성일 : 2025.03.11
- 조회수 : 1,466
(photo = prodizen)
[Medical Today = Reporter Shin Chang-ho] Prodigen, a cell therapy company, announced that it will participate in the "2025 Korean Society of Nutrition Medicine Spring Conference" to be held at the Convention Hall of the Kim Koo Baekbeom Memorial Hall in Seoul on the 16th.
The Korean Academy is an institution for education and clinical studies for doctors.It is sold out nutritional research on medical research on medical studies that affect diseases and healing, and healing is held every year related to diseases and healing.
The Korean Society of the Korean Society of the Korean Society, which is interested in nutritional medicine, is divided into four sessions and immune △ tests through immune △ tests and immune △ tests.
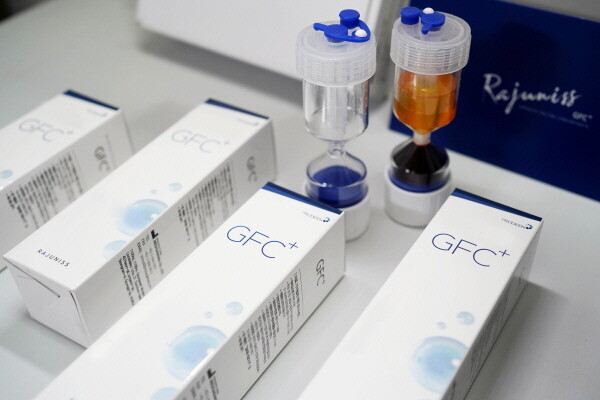
At this conference, Prodizen plans to in-depth introduce GFC+, which uses its unique plasma separation system. GFC+ is a product that can quickly and easily extract PRP, increasing the convenience of medical staff's procedures and creating a more effective treatment environment.
In particular, Rocky GFC+ is customized antibodies using various growth factors extracted from the patient's blood.It is possible to apply to a method directly injections directly to inject veins and skin and skin.Unlike existing countries centered on existing countries, it is possible to apply throughout the existing national source.In the case of vein, it is known to help through lung and liver functionality improvement and help improve the organization, and various studies are underway at school.
Prodizen has been conducted continuous research development for cell treatment and organizational renewable engineering fields.Since the first time in Korea, PRP and BMC products have been developed by developing PRP and BMC products in 2008 PRP businesses and BMC products.In particular, related faith technology technology to commercialize related faith technologies such as formal surgery, Ahn, Ahn, and mountains, etc., contributed to commercialization
Prodizen's PRP has proven its competitiveness in the global market by obtaining European CE and US FDA certification beyond Korea. It has already been recognized for its technological prowess in the United States, Canada, and China, and is also accelerating its expansion into overseas markets in Asia, Europe, and North America.
A professional official said, "We have been sold out on research development for technology advanced and accurately," said, "We have worked outstanding the productivity of professional medical staff."
Medical Today reporter Shin Chang-ho (ssangdae98@mdtoday.co.kr )








 이전글
이전글
 다음글
다음글